Mycorrhizae in a Bottle.
Can Liquid Organic Fertilisers Really Deliver VAM to Your Golf Greens?
It sounds good. But does the biology actually work that way on a putting green?
While recovering from surgery recently, I spent some time going through the literature on Arbuscular mycorrhizal fungi (AMF or VAM) in turfgrass systems, inoculant viability, and the practical realities of colonisation of mycorrhiza under intensive turf management.
The short answer is that the marketing has run a long way ahead of the science. The longer answer matters, because it involves your budget and your turf quality.
The Biology Is Real. The Products Are the Problem.
AMF form symbiotic relationships with the roots of most turfgrass. The fungus colonises the roots and then extends hyphae into the surrounding soil. The plant provides carbon. The fungus scavenges P and trace elements from soil volumes the roots can’t reach on their own. Under the right conditions, the benefits are genuine and include: better root growth, drought resistance, and nutrient uptake1Smith, S.E. & Read, D.J. (2008). Mycorrhizal Symbiosis (3rd ed.). Academic Press.2Gemma, J.N. et al. (1997a). Enhanced establishment of bentgrasses by arbuscular mycorrhizal fungi. Journal of Turfgrass Science, 73, 9–14.3Gemma, J.N. et al. (1997b). Mycorrhizal fungi improve drought resistance in creeping bentgrass. Journal of Turfgrass Science, 73, 15–29..
Nobody disputes that. The question is whether those conditions exist on your greens, and whether the products claiming to deliver these fungi actually contain them.
Why Nobody Is Checking.
Before getting into the biology, it’s worth understanding the regulatory environment these products are sold into. Because in Australia there is no federal efficacy registration for fertilisers.
Fertiliser regulation in Australia is primarily administered at state and territory level, and there is no federal pre-market efficacy registration comparable to the APVMA registration process for agricultural chemicals. Fertiliser law is a state and territory matter.
- NSW has the Fertilisers Act 1985.
- Victoria has the Agricultural and Veterinary Chemicals (Control of Use) (Fertilisers) Regulations 2015.
- Queensland regulates under the Biosecurity Regulation 2016.
But what these state regulations actually police is narrow: the maximum allowable concentrations of heavy metal like Cd, Hg, and Pb; warning statements; and minimum requirements for product descriptions on labels.
They check whether the NPK analysis on the label matches what’s in the container. They do not check whether biological claims are true.
Fertilisers are generally excluded from APVMA registration unless claims are made that trigger regulation under the Agvet Code (for example pest control or plant growth regulation claims).
So if a product is marketed as a fertiliser containing mycorrhizae, it falls through the gap. The APVMA does not touch it. The state regulators only check chemistry and contaminants, not biology.
National Code of Practice.
The industry body, Fertilizer Australia, has developed a National Code of Practice for Fertiliser Description and Labelling in conjunction with state regulators. But the Code of Practice is not underpinned by legislation. It is voluntary.
Fertilizer Australia has recently commissioned a 2025 Microbial Scoping Study for the biostimulant category, which tells you they know there’s a problem. But as of today, a manufacturer can print “contains mycorrhizae” on a liquid fertiliser label with no obligation to demonstrate that the organisms are present, viable, correctly identified to species, or functional.
This is not uniquely Australian. Koziol et al4Koziol, L., McKenna, T.P. & Bever, J.D. (2025). Meta-analysis reveals globally sourced commercial mycorrhizal inoculants fall short. New Phytologist, 246, 821–827. noted that the United States has no federal regulations governing the quality, import, or export of mycorrhizal products either. The EU is further ahead with Regulation 2019/1009, which created a distinct category for plant biostimulants including mycorrhizal fungi, with conformity assessment requirements. Australia has nothing comparable.
Keep that in mind as you read what follows. Every claim made on these product labels enters the market unchecked.
The Six Things That Would Need to Be True.
For a liquid organic fertiliser containing AMF to deliver meaningful mycorrhizal benefit on a putting green, six conditions must be met at once. Every single one. Miss any one of them and it doesn’t work.
1. They must be alive when they reach your spray tank.
AMF cannot reproduce, or even grow without a live plant root. Keeping AMF viable in a liquid formulation is a serious challenge. Even dedicated mycorrhizal inoculant companies agree that early liquid products were limited by shelf life issues (Greenhouse Grower, 2021).
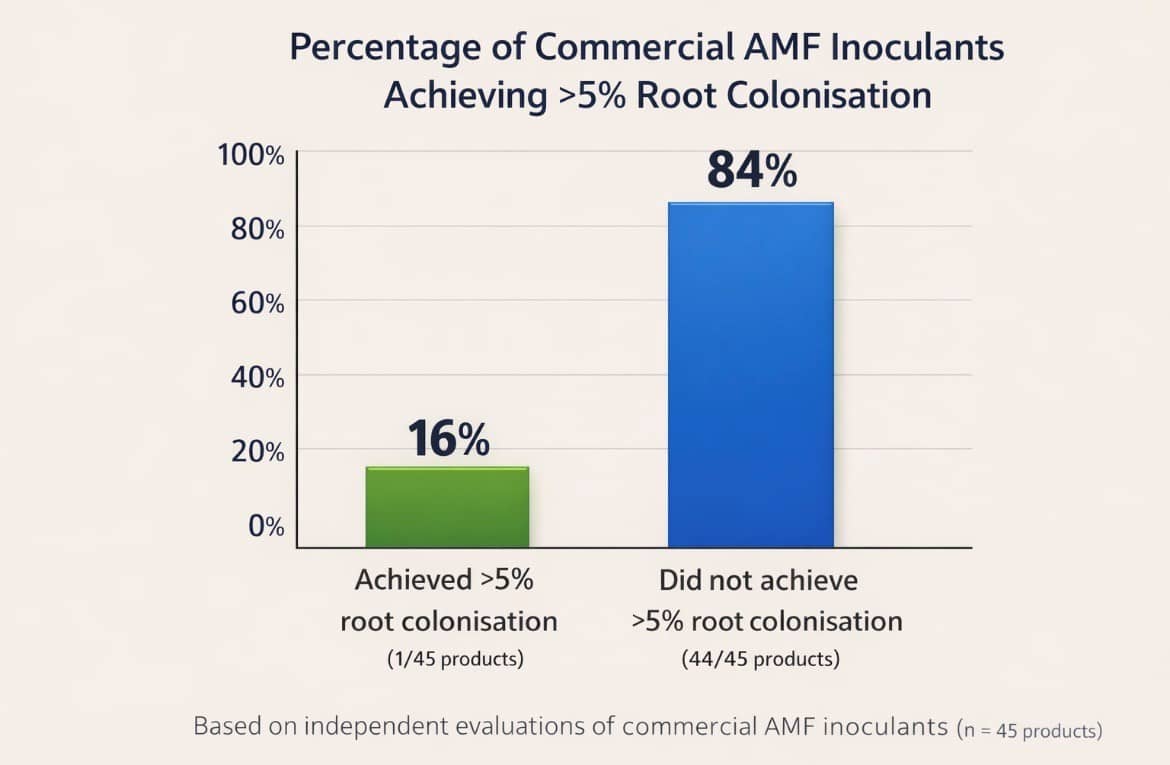
And here is where the picture gets bleak. Koziol, McKenna and Bever5Koziol, L., McKenna, T.P. & Bever, J.D. (2025). Meta-analysis reveals globally sourced commercial mycorrhizal inoculants fall short. New Phytologist, 246, 821–827. examined 302 AMF inoculants sourced globally. They found that most commercial products produce little or no root colonisation.
The commentary by Salomon6Salomon, M.J. (2025). Realising the promise of arbuscular mycorrhizal fungal biofertilisers through more applied research. New Phytologist, 246, 791–793 in the same journal described the findings as sobering, noting that the majority of commercial inoculants resulted in minimal root colonisation.
These were dedicated mycorrhizal products. Not fertilisers with mycorrhizae added as a secondary ingredient.
If purpose built inoculants fail to colonise roots, what realistic chance does a liquid organic fertiliser with some AMF spores stirred into the mix have?
Earlier work by Tarbell and Koske7Tarbell, T.J. & Koske, R.E. (2007). Evaluation of commercial arbuscular mycorrhizal inocula in a sand/peat medium. Mycorrhiza, 18, 51–56., in USGA-specification sand/peat media, found that only three out of eight products produced any mycorrhizal colonisation at the recommended use rate. Colonisation levels ranged from 0.4 to 8%.
Koziol et al.8Koziol, L., Lubin, T. & Bever, J.D. (2024). An assessment of twenty-three mycorrhizal inoculants reveals limited viability of AM fungi, pathogen contamination, and negative microbial effect on crop growth for commercial products. Applied Soil Ecology, 202, 105559. later assessed 23 mycorrhizal products and found pathogen contamination, crop mortality, and limited viability with the majority of products tested. This pattern has been repeatedly observed.
Australian Evidence.
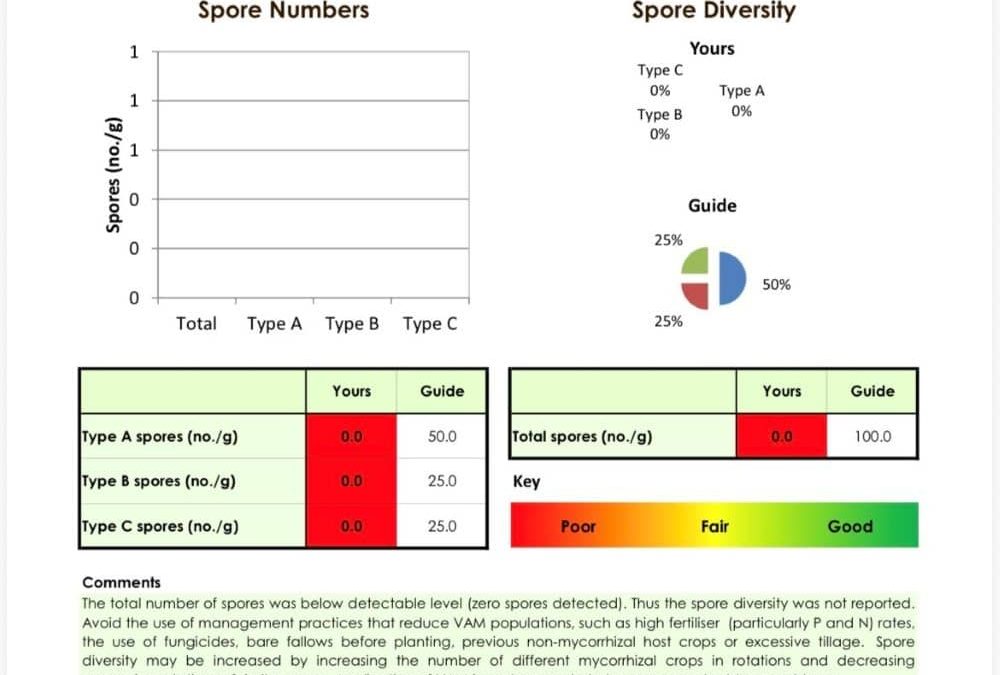
We have local data on this. In Australia, a liquid fertiliser sold for turf use and marketed as containing VAM was independently tested by two laboratories in 2021.
The Australian Genome Research Facility ran ITS diversity profiling on it. They found 35 fungal taxa. Not one of them was a mycorrhizal fungus. In fact it contained significant quantities of sourdough and wine yeast. The stuff you’d find in a bakery, not in a root cortex (AGRF Diversity Profiling Report, 2021).
Microbiology Laboratories Australia ran a separate VAM Wise spore count on a product from the same supplier. Total spores detected: zero (Microbiology Laboratories Australia, 2021).
Two labs, two methods, same answer. The product contained no mycorrhiza. What it contained was yeasts. That’s the Koziol pattern playing out in Australian products.
Why Liquid Formulations Are Problematic.
There is a key reason why liquids are a bad idea for AMF, and it goes beyond quality control failures.
When you put AMF in an aqueous suspension it faces microbial contamination, osmotic stress, oxygen limitation, and physical damage to their hyphae.
Koziol et al.9Koziol, L., Lubin, T. & Bever, J.D. (2024). An assessment of twenty-three mycorrhizal inoculants reveals limited viability of AM fungi, pathogen contamination, and negative microbial effect on crop growth for commercial products. Applied Soil Ecology, 202, 105559. noted that approximately 98% of propagules listed on commercial products are hyphal fragments. These AMF hyphae are delicate, which makes them vulnerable to damage.
The peer-reviewed literature on AMF preservation is clear about what works:
- Cold storage at 4°C10Juge, C., Samson, J., Bastien, C., Vierheilig, H., Coughlan, A. & Piché, Y. (2002). Breaking dormancy in spores of the arbuscular mycorrhizal fungus Glomus intraradices: a critical cold-storage period. Mycorrhiza, 12, 37–42., cryopreservation below −130°C11Lalaymia, I., Cranenbrouck, S. & Declerck, S. (2014). Maintenance and preservation of ectomycorrhizal and arbuscular mycorrhizal fungi. Mycorrhiza, 24, 323–337. or
- Encapsulation in polymer matrices12Vassilev, N., Vassileva, M., Azcon, R. & Medina, A. (2005). Polymer-based preparation of soil inoculants: applications to arbuscular mycorrhizal fungi. Reviews in Environmental Science and Bio/Technology, 4, 75–85..
No liquid organic fertiliser on the Australian market uses polymer encapsulation for its claimed mycorrhizal component.
The pH Preservation Claim.
At least one Australian manufacturer has also made claims that if you adjust the pH of its liquid product it puts the spores to sleep, and preserves them until you spray them on.
The idea is that if you make the liquid more acidic or alkaline it stops the spores from germinating. They then stay alive until they reach the soil. It sounds plausible. It isn’t.
pH does affect whether spores germinate. Hepper13Hepper, C.M. (1984). Regulation of spore germination of the vesicular-arbuscular mycorrhizal fungus Acaulospora laevis by soil pH. Transactions of the British Mycological Society, 83, 154–156. showed that spores of one AMF species would only germinate within a certain pH range.
Move the pH outside that range and germination slows or stops. But stopping a spore from germinating is not the same as keeping it alive.
Dormancy in mycorrhiza is a natural state the fungus controls itself14Tommerup, I.C. (1983). Spore dormancy in vesicular-arbuscular mycorrhizal fungi. Transactions of the British Mycological Society, 81, 37–45.. It is not something you can switch on if you change the acidity of the liquid around it.
No published study has shown that if you adjust the pH of a liquid product, it keeps AMF spores viable during storage on a warehouse shelf.
In fact, the research points the other way. In acidic media low pH has been shown to damage spore germination, and reduce colonisation15Jansa, J., Erb, A., Oberholzer, H.-R., Šmilauer, P. & Egli, S. (2014). Soil and geography are more important determinants of indigenous arbuscular mycorrhizal communities than management practices in Swiss agricultural soils. Molecular Ecology, 23, 2118–2135.16Ma, N., Yokoyama, K. & Marumoto, T. (2006). Stimulatory effect of peat on spore germination and hyphal growth of arbuscular mycorrhizal fungus Gigaspora margarita. Soil Science and Plant Nutrition, 52, 168–176..
If a manufacturer claims their pH preserves the fungi in the bottle, they should be able to show you colonisation data for the product at different storage intervals. How many viable spores at one month, three months, six months? Ask for it.
2. They must survive application and reach the root zone.
A liquid fertiliser is sprayed onto the turf canopy and watered in. On a USGA sand rootzone, this solution moves rapidly through the profile. The hydraulic conductivity of a properly constructed green is designed to be 150 to 300 mm per hour. Water applied to the surface drains through quickly.
AMF spores need direct physical contact with an active root to germinate and begin colonisation. In a rapidly draining sand profile, a dilute spore suspension washed in with irrigation has a transit time through the root zone measured in minutes.
The probability of a spore landing adjacent to an active root, in the right orientation, before being flushed past it and into the drainage layer, is low.
Koske and Gemma’s work showed that USGA greens are low in AMF populations because the sand medium is hostile to natural colonisation. Their 1997 survey found that golf greens constructed to USGA standards lack mycorrhizal fungi at establishment and populations are slow to increase17Koske, R.E., Gemma, J.N. & Jackson, N. (1997). Arbuscular mycorrhizal fungi associated with three species of turfgrass. Canadian Journal of Botany, 75, 320–332..
3. The Roots must be free of systemic fungicide.
This is where the whole proposition falls apart for most intensively managed greens.
On a bentgrass or ultradwarf couch golf green, the use of preventive fungicide is common in the growing season. Applications typically occur on 14 to 28 day intervals. The chemistries most commonly used for key diseases (DMI and SDHI fungicides) are systemic. They accumulate in plant tissue, and this includes the roots.
Lebanon Turf’s compilation of published fungicide-AMF interactions states plainly: roots of treated plants are not susceptible to colonisation by mycorrhizal fungi for up to three weeks after systemic fungicide treatment.
On a green that gets preventive applications every two to three weeks, there is no window when roots are receptive to new AMF colonisation.
This inhibition is fungistatic, not fungicidal. This means that AMF spores can sit tight and germinate later once the chemistry clears. But on a green that gets fresh fungicide every two to three weeks, that recovery window never occurs.

Fungicide use on a 21 day cycle never gives VAM or AMF a window to colonise roots.
Fungicide Research.
Edlinger et al18Edlinger, A. et al. (2022). Agricultural management and pesticide use reduce the functioning of beneficial plant symbionts. Nature Ecology and Evolution, 6, 1145–1154. examined AMF levels across European farmland and grassland soils. They found that fungicides had the largest impact on P transfer.
Agricultural soils with regular fungicide inputs had lower AMF levels and symbiotic function compared to unmanaged grasslands. Golf greens typically use fungicides at a much higher frequency than most arable cropping systems.
Interestingly, Bary et al19Bary, F., Gange, A.C., Crane, M. & Hagley, K.J. (2005). Fungicide levels and arbuscular mycorrhizal fungi in golf putting greens. Journal of Applied Ecology, 42, 171–180. investigated fungicide levels and AMF colonisation in golf greens in southeast England.
They measured heavy metal residues from historical fungicide use (As, Cd, Cu, Pb) and levels of three common modern fungicides (chlorothalonil, iprodione, fenarimol).
They found virtually no relationship between any chemical and AMF levels, and that there was no effect of any fungicide on AMF colonisation.
Their conclusion was that fungicides were not the main explanation for the low AMF levels. Instead, they pointed to high soil P and the extreme intensity of mowing (4.5 mm daily cuts year-round), which reduces the plant’s capacity to allocate carbon to the symbiosis.
However, that finding doesn’t let fungicides off the hook. An in vitro study from Calonne et al20Calonne, M. et al. (2012). Propiconazole inhibits the sterol 14 alpha-demethylase in Glomus irregulare like in phytopathogenic fungi. Chemosphere, 87, 376–383. shows that direct toxicity is real at the cellular level.
What they demonstrated is that on an established green, thatch is likely to degrade fungicides before they reach the roots in sufficient concentration. In fact this adds two more items to the list of reasons why golf greens are hostile to AMF.
- The P load and
- the constant defoliation from mowing. The six conditions in this article are not independent. They compound.
You cannot spray fungicide onto your greens every fortnight and expect mycorrhizal colonisation from a liquid fertiliser. These management conditions make AMF colonisation highly unlikely.
4. Soil phosphorus must be low enough to trigger the symbiosis.
The mycorrhizal symbiosis exists because both partners benefit. The plant provides carbon. The fungus provides P and trace elements. When soil P is abundant, the plant has less need for the fungal partnership and reduces the amount of carbon available to mycorrhiza. This is well established across hundreds of studies.
Even the AMF manufacturers acknowledge it. High rates of fertilisers, especially P, inhibit the formation of mycorrhizae (AgBio Inc.; Mycorrhizal Applications Inc.).
On golf greens, Mehlich-3 P levels are typically in the sufficient to high range. MLSN guidelines recommend maintaining P above 21 ppm. Many greens, particularly those with histories of granular starter fertiliser applications or organic topdressings, carry P levels well above 40 ppm. At those concentrations, the plant’s incentive to support mycorrhiza is minimal.
And here’s the contradiction at the heart of the “liquid organic fertiliser with VAM” claim. Organic fertilisers themselves typically carry meaningful P loads. You are simultaneously adding the organism and suppressing its reason for existing.
5. Soil pH must be in the right range for the specific AMF species.
Different AMF species favour different soil pH levels. Some thrive in acidic soils, and others prefer near-neutral pH soils. The species composition of AMF communities shifts with soil pH.
For a commercial product to work, the species must be compatible with the pH of your rootzone. Most liquid organic fertiliser labels (if they list species at all) include a generic mix of Glomus species. Whether those specific species are suited to your sand rootzone at its particular pH is an unknown that nobody tests.
6. The hyphal network must survive cultivation.
AMF function via a hyphal network that extends from roots out into the surrounding soil. This network is the means by which the fungus delivers nutrients.
On a golf green, you destroy that network every time you aerate or verticut. Core aeration plus verticutting, and sand topdressing, means that you constantly sever or bury the hyphal network.
In natural grasslands, the network is persistent and continuous. This contrasts with a golf green, where it must be rebuilt every time you cultivate. This is assuming the root fragments survive, and all the conditions above are all still met.
Intensive cultivation resets the clock on mycorrhizal establishment. This is another reason there are low AMF levels in maintained turf even when no fungicide pressure exists.
There is a related mechanism at work apart from this physical disruption. AMF depend on the host plant to supply carbon. On a golf green cut at 4 to 5 mm, the plant’s ability to photosynthesise is reduced.
Bary et al21Bary, F., Gange, A.C., Crane, M. & Hagley, K.J. (2005). Fungicide levels and arbuscular mycorrhizal fungi in golf putting greens. Journal of Applied Ecology, 42, 171–180. identified this mowing intensity as a likely explanation for low AMF levels in golf greens. They argued that such constant defoliation reduces the plant’s ability to move carbon to the mycorrhiza (see also Jakobsen, Smith & Smith22Jakobsen, I., Smith, S.E. & Smith, F.A. (2002). Function and diversity of arbuscular mycorrhizae in carbon and mineral nutrition. Mycorrhizal Ecology (eds M.G.A. van der Heijden & I.R. Sanders), pp. 75–92. Springer-Verlag, Berlin.. The fungus basically starves.
Cultivation destroys the network whilst mowing starves it metabolically. Both operate simultaneously on every green.
Agricultural research tells the same story. Tillage that turns the soil destroys the hyphal network and reduces AMF in subsequent crops (Hage-Ahmed et al23Hage-Ahmed, K., Rosner, K. & Steinkellner, S. (2018). Arbuscular mycorrhizal fungi and their response to pesticides. Pest Management Science. DOI: 10.1002/ps.5220..
Green’s cultivation disturbs the surface soil more frequently than most agricultural tillage systems. More frequent, shallower, and targeted right where the active network is concentrated.
What the Field Trials Show.
Beyond laboratory analyses, there is Australian field trial data on the performance of VAM containing liquid fertilisers used on golf greens under real management conditions.
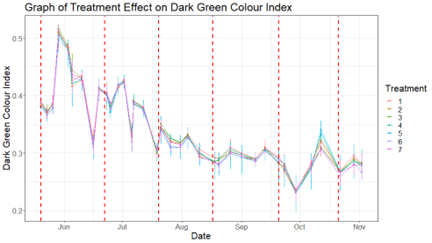
In 2019, Jerry Spencer and Cameron Smith ran field trial at Bonnie Doon Golf Club in Sydney. Randomised block design, 60 plots, four replicates per treatment, monthly applications at label rate, DGCI measured by digital image analysis, statistics done in R. The trial ran for over 100 days on a mixed Poa annua/Agrostis practice putting green24Spencer, J. & Smith, C. (2019). Bonnie Doon Trial Report Summary: Impact of Organic/Biological Amendments on Dark Green Colour Index (DGCI). Bonnie Doon Golf Club, August 2019.
The VAM-containing liquid was a high P product marketed with VAM organics. It produced no statistically significant improvement over the untreated control. Not at any measurement point. Not once across the entire trial.
The authors’ conclusion: the results did not warrant standalone usage if the aim was to improve turf quality.
The two best-performing treatments in the same trial were a concentrated silicon product at 25% SiO₂, and a combination of Marmite and Berocca tablets, delivering B vitamins, yeast extract, and folic acid.
Both were statistically better than the control on multiple dates. A silicon product and something you could buy at Woolworths outperformed a product built around mycorrhizal claims.
The authors noted the VAM product might have done better with supplementary nutrition. But the trial was properly designed, properly analysed, and its conclusions line up with everything else in this article.
The Poa annua Question.
One of the most frequently cited reasons for the use of AMF products on golf greens comes from the work of Gange, Lindsay and Ellis.25Gange, A.C., Lindsay, D.E. & Ellis, L.S. (1999). Can arbuscular mycorrhizal fungi be used to control the undesirable grass Poa annua on golf courses? Journal of Applied Ecology, 36, 909–919.
They found that on UK golf courses, greens with higher AMF levels had less Poa annua and more Agrostis stolonifera. The proposed mechanism was that AMF act as a carbon drain on Poa annua, reducing its growth while benefiting bentgrass. This finding has been heavily promoted by mycorrhizal product manufacturers.
This is correct but the cause needs scrutiny. Greens with higher AMF levels in Gange’s study were also greens with lower fungicide inputs, lower fertility, and less intensive management. Those same management factors independently favour bentgrass over Poa annua.
The correlation between AMF and Poa annua suppression may reflect less intensive management, but this is not a direct biological mechanism.
Aamlid and Andersen26Aamlid, T.S. & Andersen, T.E. (2012). Composted garden waste as organic amendment to the USGA-rootzone and topdressing sand on red fescue (Festuca rubra) greens. International Turfgrass Society Research Journal. found that mycorrhizal colonisation was actually slightly but significantly higher in Poa annua than in red fescue on their research greens.
In compost-amended rootzones, with higher AMF populations, there was worse Poa annua encroachment than on peat-amended rootzones. This directly contradicts the simple narrative that more AMF equals less Poa annua.
The honest conclusion is that the relationship between AMF and Poa annua in golf greens is possibly confounded by management intensity, and certainly not strong enough to justify purchasing a product on that basis alone.
The Quality Control Problem.
Even setting aside the hostile environment of a golf green, there is a big problem with the products themselves.
- The Koziol et al27Koziol, L., McKenna, T.P. & Bever, J.D. (2025). Meta-analysis reveals globally sourced commercial mycorrhizal inoculants fall short. New Phytologist, 246, 821–827. found that the global commercial AMF inoculant industry, is delivering products that mostly do not work.
- Tarbell and Koske28Tarbell, T.J. & Koske, R.E. (2007). Evaluation of commercial arbuscular mycorrhizal inocula in a sand/peat medium. Mycorrhiza, 18, 51–56. found that five of eight dedicated commercial inocula failed to colonise roots in USGA-spec media.
- Boussageon et al.29Boussageon, R. et al. (2025). Poor quality of commercial arbuscular mycorrhizal inoculants used for agriculture and home gardening. Journal of Sustainable Agriculture and Environment. confirmed that the majority of commercial products fail to facilitate even minimal root colonisation.
The Australian testing tells the same story.
- ITS sequencing by the Australian Genome Research Facility on a liquid fertiliser sold as containing VAM, found 35 fungal taxa and zero mycorrhizal fungi. The dominant organisms were yeasts.
- A separate spore count by Microbiology Laboratories Australia on a product from the same supplier found zero spores across all types (AGRF, 2021; Microbiology Laboratories Australia, 2021).
These are products whose entire purpose is to deliver viable AMF. A liquid organic fertiliser that lists “contains mycorrhizae” on the label, with no species identification, no propagule count, no viability data, and no independent testing, is even less likely to work.
Ask the manufacturer three questions:
- What AMF species are included?
- How many viable propagules per millilitre at the point of sale?
- What independent colonisation data exists for this product in sand-based rootzones under fungicide programmes?
If they cannot answer all three, you are buying a marketing claim, not a biological input.
So Where Does AMF Inoculation Actually Make Sense in Turf?
The research supports AMF benefits in specific, limited contexts.
Establishment.
Gemma et al30Gemma, J.N. et al. (1997a). Enhanced establishment of bentgrasses by arbuscular mycorrhizal fungi. Journal of Turfgrass Science, 73, 9–14.31Gemma, J.N. et al. (1997b). Mycorrhizal fungi improve drought resistance in creeping bentgrass. Journal of Turfgrass Science, 73, 15–29. showed genuine improvements in bentgrass establishment and early drought resistance when AMF inoculant was incorporated into the rootzone at seeding.
This is the best-case scenario: new sand medium with no competing native AMF, no fungicide residues, no established hyphal network to displace, and direct incorporation of granular inoculant into the root zone. If you are building a new green or doing a complete renovation, incorporating a dedicated, quality-tested AMF inoculant during construction has reasonable scientific support.
Low-input turf.
Fairways, roughs, and naturalized areas that receive minimal fungicide, moderate fertility, and infrequent cultivation are environments where AMF can establish and persist. These are also the areas where the benefits of improved nutrient scavenging and drought tolerance matter most, because the turf is expected to perform with fewer inputs.
Greens managed without fungicides.
In Scandinavia, the UK, and parts of northern Europe, some courses manage fine fescue greens with very low fungicide input. These greens are the closest analogue to the conditions under which AMF research has shown benefits. If you’re managing a links-style course on fescue greens with minimal chemistry, AMF inoculation during renovation or aeration may have merit.
Early trial work in Australia with a dedicated granular AMF product (Glomus iranicum var. tenuihypharum) applied to a mixed Poa/Agrostis sward showed directional improvements in root length, root width, NDVI, and the number of connected root components. 32Spencer, J. (2019b). Biologicals in Rootzone Media. Gilba Solutions Pty Ltd, February 2019. The results were not statistically significant, but the trend was consistent across all measured parameters. That’s what you’d expect from a properly formulated product applied directly to the rootzone in a context where the biology has a chance to establish. It’s also a long way from what a liquid foliar spray can deliver.
None of these scenarios involve spraying a liquid organic fertiliser onto a bentgrass or ultradwarf couch green that’s receiving biweekly fungicide applications.
What Should You Actually Do?
If you want to improve the biological health of your rootzone, the evidence points toward a different set of actions than spraying liquid organics with mycorrhiza claims.
Manage organic matter properly.
A rootzone with appropriate organic matter levels (commonly cited targets for sand-based greens are roughly 2–4% organic matter in the upper surface layer.) supports a diverse microbial community.
Too much OM creates anaerobic conditions, soft surfaces, and disease pressure. Too little leaves the microbial community without substrate. The balance matters more than any inoculant.
Reduce Fungicide where you can, not where you can’t.
If your greens require preventive fungicides for disease control, that’s a management reality. Pouring money into mycorrhizal products that will be killed by your next fungicide application is waste of $. Put those dollars into better soil testing, more precise fertility, or improve your drainage instead.
Use dedicated, quality-tested AMF products in the right context.
If you’re renovating or build new greens, use granular AMF inoculants from reputable suppliers (with documented species, propagule counts, and colonisation data) in construction. This is the one context where the science supports the practice.
Don’t confuse “natural” with “effective.”
The word “organic” on a fertiliser label tells you nothing about whether the product will improve your turf. And the word “mycorrhiza” on a liquid fertiliser label, without any supporting data, tells you even less.
The biology of AMF is fascinating and genuinely important to plant nutrition in natural and low-input systems. The science is not in dispute. What is in dispute is the claim that you can deliver this biology in a spray bottle and it will function on a surface that is engineered, cultivated, and chemically managed in ways that are hostile to fungal colonisation.
The golf green is one of the most intensively managed plant communities on earth. It is the worst possible environment to establish mycorrhizae, and the products that claim to do it have not demonstrated that they can.
Spend your money on what the evidence says works. Test your soil. Manage your organic matter. Match your N source to your disease profile. And if someone tells you their liquid fertiliser will build mycorrhizae on your bentgrass greens while you spray propiconazole every three weeks, ask them to show you the colonisation data.
They won’t have it.
References
Senior Turf Agronomist, Gilba Solutions Pty Ltd
Hons Soil Science (Newcastle Upon Tyne). Former STRI agronomist. 35+ years’ experience in turf agronomy, fertiliser programs, and weed management across Australia.